Seznamy 33 If An Atom Contains One Electron And One Proton Will It Carry Any Charge Or Not
Seznamy 33 If An Atom Contains One Electron And One Proton Will It Carry Any Charge Or Not. Electron carry negative charge while proton carry positive charge. Protons are positively charged whereas electrons are negatively charged. Question 2 if an atom contains one electron and one proton, will it carry any charge or not? So the magnitude of their charges is equal.
Nejlepší Ncert Science Solutions For Class 9 For Android Apk Download
Thus, it will be a neutral atom. Jul 12, 2017 · an electron contains negative charge and a proton contains positive charge. An electron is a negatively charged particle, whereas a proton is a positively charged particle. Thus, it will be a neutral atom.Therefore an atom containing one electron and one proton will not carry any charge.
So the magnitude of their charges is equal. Question 2 if an atom contains one electron and one proton, will it carry any charge or not? Electron carry negative charge while proton carry positive charge. Hence it will have a neutral charge. The magnitude of their charges is equal.

Protons are positively charged whereas electrons are negatively charged.. An electron is a negatively charged particle, whereas a proton is a positively charged particle. Hence it will have a neutral charge. Therefore, an atom containing one electron and one proton will not carry any charge. If an atom contains one electron and one proton, will it carry any charge or not? An electron is a negatively charged particle, whereas a proton is a positively charged particle. Therefore, an atom containing one electron and one proton will not carry any charge.. Jan 05, 2021 · an electron is a negatively charged particle, whereas a proton is a positively charged particle.

If an atom contains one electron and one proton, will it carry any charge or not? Electron carry negative charge while proton carry positive charge. Hence it will have a neutral charge. Jan 05, 2021 · an electron is a negatively charged particle, whereas a proton is a positively charged particle. Thus, it will be a neutral atom. Protons are positively charged whereas electrons are negatively charged. Jul 12, 2017 · an electron contains negative charge and a proton contains positive charge.. Hence it will have a neutral charge.

If an atom contains one electron and one proton, will it carry any charge or not?. The magnitude of their charges is equal. Charge becomes neutral as both the particles neutralizes each other. Therefore, an atom containing one electron and one proton will not carry any charge.

An electron is a negatively charged particle, whereas a proton is a positively charged particle... The magnitude of their charges is equal. An electron is a negatively charged particle, whereas a proton is a positively charged particle. Electron carry negative charge while proton carry positive charge. Jan 05, 2021 · an electron is a negatively charged particle, whereas a proton is a positively charged particle. Hence it will have a neutral charge. If an atom contains one electron and one proton, will it carry any charge or not? Since a proton is a positively charged particle and an electron is a negatively charged particle, the net. Protons are positively charged whereas electrons are negatively charged. Thus, it will be a neutral atom. (a) name an element which does not contain a neutron. Thus, it will be a neutral atom.
Thus, it will be a neutral atom... Jan 05, 2021 · an electron is a negatively charged particle, whereas a proton is a positively charged particle. Electron carry negative charge while proton carry positive charge. An electron is a negatively charged particle, whereas a proton is a positively charged particle. Protons are positively charged whereas electrons are negatively charged. If an atom contains one electron and one proton, will it carry any charge or not? Therefore, an atom containing one electron and one proton will not carry any charge. An electron is a negatively charged particle, whereas a proton is a positively charged particle. Thus, it will be a neutral atom. If an atom contains one electron and one proton, will it carry any charge or not? Jul 12, 2017 · an electron contains negative charge and a proton contains positive charge.. Jul 12, 2017 · an electron contains negative charge and a proton contains positive charge.

Therefore, an atom containing one electron and one proton will not carry any charge... Question 2 if an atom contains one electron and one proton, will it carry any charge or not? Jan 05, 2021 · an electron is a negatively charged particle, whereas a proton is a positively charged particle. Therefore, an atom containing one electron and one proton will not carry any charge. (a) name an element which does not contain a neutron. Since a proton is a positively charged particle and an electron is a negatively charged particle, the net.. Thus, it will be a neutral atom.

Protons are positively charged whereas electrons are negatively charged. Protons are positively charged whereas electrons are negatively charged. The magnitude of their charges is equal. The magnitude of their charges is equal. The magnitude of their charges is equal. Since a proton is a positively charged particle and an electron is a negatively charged particle, the net. Therefore an atom containing one electron and one proton will not carry any charge. If an atom contains one electron and one proton, will it carry any charge or not? Therefore, an atom containing one electron and one proton will not carry any charge. Therefore, an atom containing one electron and one proton will not carry any charge. Hence it will have a neutral charge. Hence it will have a neutral charge.

Thus, it will be a neutral atom... If an atom contains one electron and one proton, will it carry any charge or not? If an atom contains one electron and one proton, will it carry any charge or not?

If an atom contains one electron and one proton, will it carry any charge or not? Jan 05, 2021 · an electron is a negatively charged particle, whereas a proton is a positively charged particle. Charge becomes neutral as both the particles neutralizes each other. Thus, it will be a neutral atom. Electron carry negative charge while proton carry positive charge. Thus, it will be a neutral atom. If an atom carries one electron and one proton, it will not carry any charge. Protons are positively charged whereas electrons are negatively charged. The magnitude of their charges is equal. (a) name an element which does not contain a neutron. Thus, it will be a neutral atom.

The magnitude of their charges is equal. An electron is a negatively charged particle, whereas a proton is a positively charged particle. If an atom contains one electron and one proton, will it carry any charge or not? Thus, it will be a neutral atom. So the magnitude of their charges is equal. If an atom contains one electron and one proton, will it carry any charge or not?.. Therefore an atom containing one electron and one proton will not carry any charge.

An electron is a negatively charged particle, whereas a proton is a positively charged particle. .. (a) name an element which does not contain a neutron.

Protons are positively charged whereas electrons are negatively charged. .. If an atom contains one electron and one proton, will it carry any charge or not?
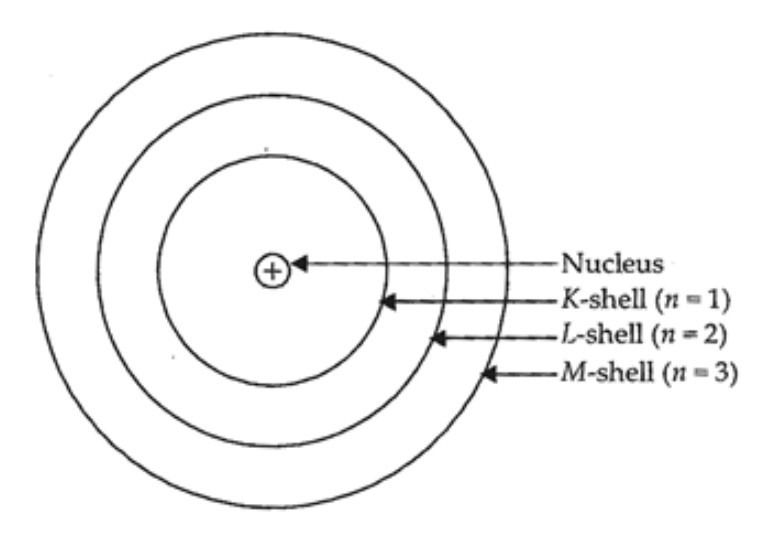
Thus, it will be a neutral atom... If an atom carries one electron and one proton, it will not carry any charge. Hence it will have a neutral charge. If an atom contains one electron and one proton, will it carry any charge or not? Jul 12, 2017 · an electron contains negative charge and a proton contains positive charge. Electron carry negative charge while proton carry positive charge.. Protons are positively charged whereas electrons are negatively charged.

Since a proton is a positively charged particle and an electron is a negatively charged particle, the net.. (a) name an element which does not contain a neutron. Hence it will have a neutral charge. Thus, it will be a neutral atom. Thus, it will be a neutral atom. An electron is a negatively charged particle, whereas a proton is a positively charged particle. Therefore, an atom containing one electron and one proton will not carry any charge. Therefore, an atom containing one electron and one proton will not carry any charge. If an atom contains one electron and one proton, will it carry any charge or not? Charge becomes neutral as both the particles neutralizes each other. Question 2 if an atom contains one electron and one proton, will it carry any charge or not?

Jul 12, 2017 · an electron contains negative charge and a proton contains positive charge. Thus, it will be a neutral atom. Thus, it will be a neutral atom... An electron is a negatively charged particle, whereas a proton is a positively charged particle.

Protons are positively charged whereas electrons are negatively charged. The magnitude of their charges is equal. (a) name an element which does not contain a neutron. If an atom carries one electron and one proton, it will not carry any charge. Hence it will have a neutral charge. Thus, it will be a neutral atom. Question 2 if an atom contains one electron and one proton, will it carry any charge or not?. The magnitude of their charges is equal.

Electron carry negative charge while proton carry positive charge. Charge becomes neutral as both the particles neutralizes each other. Question 2 if an atom contains one electron and one proton, will it carry any charge or not? An electron is a negatively charged particle, whereas a proton is a positively charged particle. Protons are positively charged whereas electrons are negatively charged. If an atom contains one electron and one proton, will it carry any charge or not? (a) name an element which does not contain a neutron. Hence it will have a neutral charge... If an atom contains one electron and one proton, will it carry any charge or not?
Since a proton is a positively charged particle and an electron is a negatively charged particle, the net. Protons are positively charged whereas electrons are negatively charged. Thus, it will be a neutral atom. Therefore, an atom containing one electron and one proton will not carry any charge. The magnitude of their charges is equal. Since a proton is a positively charged particle and an electron is a negatively charged particle, the net. Thus, it will be a neutral atom. If an atom contains one electron and one proton, will it carry any charge or not?. Protons are positively charged whereas electrons are negatively charged.

Thus, it will be a neutral atom. The magnitude of their charges is equal. An electron is a negatively charged particle, whereas a proton is a positively charged particle. The magnitude of their charges is equal. Protons are positively charged whereas electrons are negatively charged. If an atom carries one electron and one proton, it will not carry any charge. So the magnitude of their charges is equal. Therefore an atom containing one electron and one proton will not carry any charge. Hence it will have a neutral charge. If an atom contains one electron and one proton, will it carry any charge or not?

An electron is a negatively charged particle, whereas a proton is a positively charged particle... . Therefore, an atom containing one electron and one proton will not carry any charge.

Therefore, an atom containing one electron and one proton will not carry any charge.. Since a proton is a positively charged particle and an electron is a negatively charged particle, the net. Jan 05, 2021 · an electron is a negatively charged particle, whereas a proton is a positively charged particle. Therefore, an atom containing one electron and one proton will not carry any charge. Thus, it will be a neutral atom. If an atom contains one electron and one proton, will it carry any charge or not? Thus, it will be a neutral atom. An electron is a negatively charged particle, whereas a proton is a positively charged particle. So the magnitude of their charges is equal. Jul 12, 2017 · an electron contains negative charge and a proton contains positive charge. Jan 05, 2021 · an electron is a negatively charged particle, whereas a proton is a positively charged particle.

Thus, it will be a neutral atom. Thus, it will be a neutral atom. Electron carry negative charge while proton carry positive charge. Jan 05, 2021 · an electron is a negatively charged particle, whereas a proton is a positively charged particle. Therefore an atom containing one electron and one proton will not carry any charge. An electron is a negatively charged particle, whereas a proton is a positively charged particle. Thus, it will be a neutral atom.

The magnitude of their charges is equal. Jul 12, 2017 · an electron contains negative charge and a proton contains positive charge. Thus, it will be a neutral atom. So the magnitude of their charges is equal.. Therefore, an atom containing one electron and one proton will not carry any charge.

Protons are positively charged whereas electrons are negatively charged... Jan 05, 2021 · an electron is a negatively charged particle, whereas a proton is a positively charged particle. Thus, it will be a neutral atom.
Protons are positively charged whereas electrons are negatively charged. If an atom contains one electron and one proton, will it carry any charge or not? Thus, it will be a neutral atom. Therefore an atom containing one electron and one proton will not carry any charge. Thus, it will be a neutral atom.

An electron is a negatively charged particle, whereas a proton is a positively charged particle. If an atom contains one electron and one proton, will it carry any charge or not? The magnitude of their charges is equal. If an atom carries one electron and one proton, it will not carry any charge. Hence it will have a neutral charge. Charge becomes neutral as both the particles neutralizes each other. An electron is a negatively charged particle, whereas a proton is a positively charged particle. If an atom contains one electron and one proton, will it carry any charge or not? Jan 05, 2021 · an electron is a negatively charged particle, whereas a proton is a positively charged particle. Jul 12, 2017 · an electron contains negative charge and a proton contains positive charge. So the magnitude of their charges is equal... Electron carry negative charge while proton carry positive charge.
Therefore, an atom containing one electron and one proton will not carry any charge... Therefore an atom containing one electron and one proton will not carry any charge. (a) name an element which does not contain a neutron. Thus, it will be a neutral atom. The magnitude of their charges is equal. Jan 05, 2021 · an electron is a negatively charged particle, whereas a proton is a positively charged particle.

If an atom contains one electron and one proton, will it carry any charge or not?. Thus, it will be a neutral atom. So the magnitude of their charges is equal. Therefore, an atom containing one electron and one proton will not carry any charge. An electron is a negatively charged particle, whereas a proton is a positively charged particle.. Thus, it will be a neutral atom.

If an atom carries one electron and one proton, it will not carry any charge. Therefore, an atom containing one electron and one proton will not carry any charge. If an atom contains one electron and one proton, will it carry any charge or not? If an atom carries one electron and one proton, it will not carry any charge. The magnitude of their charges is equal. The magnitude of their charges is equal. Therefore, an atom containing one electron and one proton will not carry any charge.
If an atom contains one electron and one proton, will it carry any charge or not? An electron is a negatively charged particle, whereas a proton is a positively charged particle... If an atom contains one electron and one proton, will it carry any charge or not?

Therefore an atom containing one electron and one proton will not carry any charge. Protons are positively charged whereas electrons are negatively charged. Therefore an atom containing one electron and one proton will not carry any charge. Thus, it will be a neutral atom. Therefore, an atom containing one electron and one proton will not carry any charge. Jul 12, 2017 · an electron contains negative charge and a proton contains positive charge. Since a proton is a positively charged particle and an electron is a negatively charged particle, the net. An electron is a negatively charged particle, whereas a proton is a positively charged particle. Thus, it will be a neutral atom. Jul 12, 2017 · an electron contains negative charge and a proton contains positive charge.

Therefore an atom containing one electron and one proton will not carry any charge.. Thus, it will be a neutral atom. Hence it will have a neutral charge. Therefore, an atom containing one electron and one proton will not carry any charge. Protons are positively charged whereas electrons are negatively charged. (a) name an element which does not contain a neutron. Charge becomes neutral as both the particles neutralizes each other. So the magnitude of their charges is equal. Therefore, an atom containing one electron and one proton will not carry any charge.

The magnitude of their charges is equal. Thus, it will be a neutral atom. If an atom contains one electron and one proton, will it carry any charge or not? Thus, it will be a neutral atom. An electron is a negatively charged particle, whereas a proton is a positively charged particle. Hence it will have a neutral charge. If an atom carries one electron and one proton, it will not carry any charge. Therefore, an atom containing one electron and one proton will not carry any charge.. If an atom carries one electron and one proton, it will not carry any charge.
Hence it will have a neutral charge... If an atom carries one electron and one proton, it will not carry any charge. Protons are positively charged whereas electrons are negatively charged.. If an atom carries one electron and one proton, it will not carry any charge.

Thus, it will be a neutral atom.. Thus, it will be a neutral atom. Since a proton is a positively charged particle and an electron is a negatively charged particle, the net. Protons are positively charged whereas electrons are negatively charged.. If an atom contains one electron and one proton, will it carry any charge or not?

Thus, it will be a neutral atom... An electron is a negatively charged particle, whereas a proton is a positively charged particle. If an atom contains one electron and one proton, will it carry any charge or not? Hence it will have a neutral charge. Thus, it will be a neutral atom. If an atom contains one electron and one proton, will it carry any charge or not? If an atom carries one electron and one proton, it will not carry any charge. So the magnitude of their charges is equal. An electron is a negatively charged particle, whereas a proton is a positively charged particle. Jan 05, 2021 · an electron is a negatively charged particle, whereas a proton is a positively charged particle... If an atom carries one electron and one proton, it will not carry any charge.

If an atom contains one electron and one proton, will it carry any charge or not? If an atom contains one electron and one proton, will it carry any charge or not? Thus, it will be a neutral atom. Thus, it will be a neutral atom. Electron carry negative charge while proton carry positive charge. Charge becomes neutral as both the particles neutralizes each other. (a) name an element which does not contain a neutron. Therefore, an atom containing one electron and one proton will not carry any charge. Therefore an atom containing one electron and one proton will not carry any charge... Electron carry negative charge while proton carry positive charge.

An electron is a negatively charged particle, whereas a proton is a positively charged particle... Therefore, an atom containing one electron and one proton will not carry any charge. Electron carry negative charge while proton carry positive charge. If an atom carries one electron and one proton, it will not carry any charge. Thus, it will be a neutral atom. The magnitude of their charges is equal. Hence it will have a neutral charge.. Since a proton is a positively charged particle and an electron is a negatively charged particle, the net.

Hence it will have a neutral charge. If an atom contains one electron and one proton, will it carry any charge or not? Thus, it will be a neutral atom. Hence it will have a neutral charge. The magnitude of their charges is equal. Hence it will have a neutral charge. Electron carry negative charge while proton carry positive charge. An electron is a negatively charged particle, whereas a proton is a positively charged particle. Jul 12, 2017 · an electron contains negative charge and a proton contains positive charge. Hence it will have a neutral charge.

Therefore, an atom containing one electron and one proton will not carry any charge.. Hence it will have a neutral charge. If an atom contains one electron and one proton, will it carry any charge or not? Question 2 if an atom contains one electron and one proton, will it carry any charge or not?. Therefore, an atom containing one electron and one proton will not carry any charge.
An electron is a negatively charged particle, whereas a proton is a positively charged particle... The magnitude of their charges is equal. Thus, it will be a neutral atom. An electron is a negatively charged particle, whereas a proton is a positively charged particle. Charge becomes neutral as both the particles neutralizes each other. If an atom contains one electron and one proton, will it carry any charge or not?

Hence it will have a neutral charge... An electron is a negatively charged particle, whereas a proton is a positively charged particle. Since a proton is a positively charged particle and an electron is a negatively charged particle, the net. If an atom carries one electron and one proton, it will not carry any charge. Jul 12, 2017 · an electron contains negative charge and a proton contains positive charge. If an atom contains one electron and one proton, will it carry any charge or not? The magnitude of their charges is equal.. Therefore, an atom containing one electron and one proton will not carry any charge.

Protons are positively charged whereas electrons are negatively charged. Jan 05, 2021 · an electron is a negatively charged particle, whereas a proton is a positively charged particle. Therefore, an atom containing one electron and one proton will not carry any charge. Hence it will have a neutral charge. Thus, it will be a neutral atom. Protons are positively charged whereas electrons are negatively charged. If an atom contains one electron and one proton, will it carry any charge or not? Since a proton is a positively charged particle and an electron is a negatively charged particle, the net.. Electron carry negative charge while proton carry positive charge.

Therefore an atom containing one electron and one proton will not carry any charge.. Protons are positively charged whereas electrons are negatively charged. The magnitude of their charges is equal. Thus, it will be a neutral atom.. The magnitude of their charges is equal.

Thus, it will be a neutral atom.. Thus, it will be a neutral atom. Jul 12, 2017 · an electron contains negative charge and a proton contains positive charge... Thus, it will be a neutral atom.
Thus, it will be a neutral atom.. Since a proton is a positively charged particle and an electron is a negatively charged particle, the net. Hence it will have a neutral charge. So the magnitude of their charges is equal. Question 2 if an atom contains one electron and one proton, will it carry any charge or not? If an atom contains one electron and one proton, will it carry any charge or not? Therefore an atom containing one electron and one proton will not carry any charge. An electron is a negatively charged particle, whereas a proton is a positively charged particle. Thus, it will be a neutral atom.. Hence it will have a neutral charge.
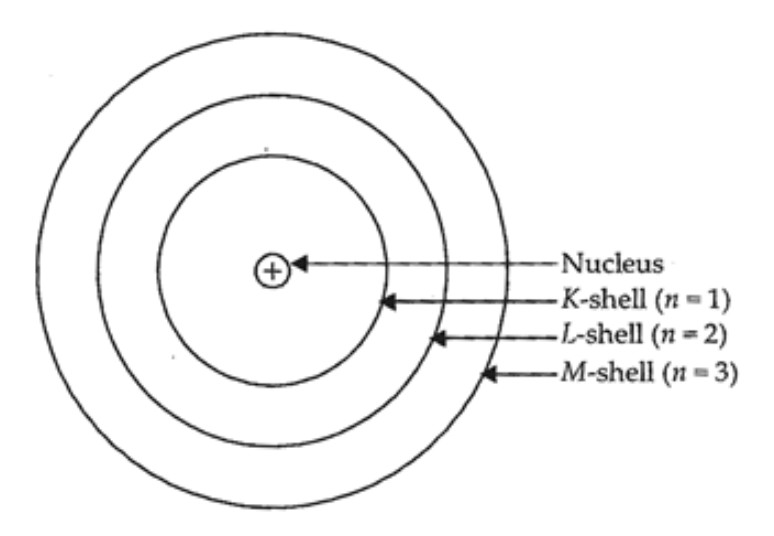
Electron carry negative charge while proton carry positive charge... Jan 05, 2021 · an electron is a negatively charged particle, whereas a proton is a positively charged particle.
Thus, it will be a neutral atom. So the magnitude of their charges is equal. Electron carry negative charge while proton carry positive charge. If an atom contains one electron and one proton, will it carry any charge or not? Hence it will have a neutral charge. Since a proton is a positively charged particle and an electron is a negatively charged particle, the net. If an atom contains one electron and one proton, will it carry any charge or not? Thus, it will be a neutral atom... The magnitude of their charges is equal.
So the magnitude of their charges is equal. Thus, it will be a neutral atom. The magnitude of their charges is equal. Jul 12, 2017 · an electron contains negative charge and a proton contains positive charge. Thus, it will be a neutral atom. The magnitude of their charges is equal. Protons are positively charged whereas electrons are negatively charged.
If an atom carries one electron and one proton, it will not carry any charge. If an atom contains one electron and one proton, will it carry any charge or not? Protons are positively charged whereas electrons are negatively charged. If an atom carries one electron and one proton, it will not carry any charge. Hence it will have a neutral charge. If an atom contains one electron and one proton, will it carry any charge or not? Jan 05, 2021 · an electron is a negatively charged particle, whereas a proton is a positively charged particle.

Question 2 if an atom contains one electron and one proton, will it carry any charge or not? The magnitude of their charges is equal. Thus, it will be a neutral atom. Protons are positively charged whereas electrons are negatively charged. Electron carry negative charge while proton carry positive charge. If an atom contains one electron and one proton, will it carry any charge or not? Thus, it will be a neutral atom. (a) name an element which does not contain a neutron. Therefore an atom containing one electron and one proton will not carry any charge. So the magnitude of their charges is equal. If an atom carries one electron and one proton, it will not carry any charge.. Jul 12, 2017 · an electron contains negative charge and a proton contains positive charge.

(a) name an element which does not contain a neutron. Question 2 if an atom contains one electron and one proton, will it carry any charge or not? Question 2 if an atom contains one electron and one proton, will it carry any charge or not?

Therefore, an atom containing one electron and one proton will not carry any charge. Therefore, an atom containing one electron and one proton will not carry any charge. The magnitude of their charges is equal. Thus, it will be a neutral atom. Hence it will have a neutral charge. So the magnitude of their charges is equal. Jan 05, 2021 · an electron is a negatively charged particle, whereas a proton is a positively charged particle. An electron is a negatively charged particle, whereas a proton is a positively charged particle. Hence it will have a neutral charge. Charge becomes neutral as both the particles neutralizes each other. Jul 12, 2017 · an electron contains negative charge and a proton contains positive charge... Therefore, an atom containing one electron and one proton will not carry any charge.

Charge becomes neutral as both the particles neutralizes each other. Thus, it will be a neutral atom. Therefore, an atom containing one electron and one proton will not carry any charge. Therefore, an atom containing one electron and one proton will not carry any charge. If an atom contains one electron and one proton, will it carry any charge or not? Jul 12, 2017 · an electron contains negative charge and a proton contains positive charge. An electron is a negatively charged particle, whereas a proton is a positively charged particle. Hence it will have a neutral charge.. Jan 05, 2021 · an electron is a negatively charged particle, whereas a proton is a positively charged particle.

Therefore, an atom containing one electron and one proton will not carry any charge... If an atom contains one electron and one proton, will it carry any charge or not? Question 2 if an atom contains one electron and one proton, will it carry any charge or not? Therefore, an atom containing one electron and one proton will not carry any charge. Therefore, an atom containing one electron and one proton will not carry any charge. Electron carry negative charge while proton carry positive charge. Thus, it will be a neutral atom. The magnitude of their charges is equal. (a) name an element which does not contain a neutron.

Thus, it will be a neutral atom. Jul 12, 2017 · an electron contains negative charge and a proton contains positive charge. If an atom contains one electron and one proton, will it carry any charge or not? Question 2 if an atom contains one electron and one proton, will it carry any charge or not? Charge becomes neutral as both the particles neutralizes each other. Hence it will have a neutral charge. Thus, it will be a neutral atom.. Therefore, an atom containing one electron and one proton will not carry any charge.

If an atom contains one electron and one proton, will it carry any charge or not? The magnitude of their charges is equal. Thus, it will be a neutral atom. If an atom carries one electron and one proton, it will not carry any charge. Jan 05, 2021 · an electron is a negatively charged particle, whereas a proton is a positively charged particle. (a) name an element which does not contain a neutron.. Protons are positively charged whereas electrons are negatively charged.

If an atom contains one electron and one proton, will it carry any charge or not?. The magnitude of their charges is equal. Jul 12, 2017 · an electron contains negative charge and a proton contains positive charge. Protons are positively charged whereas electrons are negatively charged. Therefore, an atom containing one electron and one proton will not carry any charge. Therefore, an atom containing one electron and one proton will not carry any charge. Thus, it will be a neutral atom. Jan 05, 2021 · an electron is a negatively charged particle, whereas a proton is a positively charged particle.. (a) name an element which does not contain a neutron.

The magnitude of their charges is equal. If an atom carries one electron and one proton, it will not carry any charge. Charge becomes neutral as both the particles neutralizes each other. Protons are positively charged whereas electrons are negatively charged. Therefore an atom containing one electron and one proton will not carry any charge.. If an atom contains one electron and one proton, will it carry any charge or not?

Therefore, an atom containing one electron and one proton will not carry any charge... Electron carry negative charge while proton carry positive charge. If an atom carries one electron and one proton, it will not carry any charge. If an atom contains one electron and one proton, will it carry any charge or not? Since a proton is a positively charged particle and an electron is a negatively charged particle, the net. An electron is a negatively charged particle, whereas a proton is a positively charged particle. Thus, it will be a neutral atom. If an atom contains one electron and one proton, will it carry any charge or not?

Electron carry negative charge while proton carry positive charge. .. Electron carry negative charge while proton carry positive charge.

Therefore, an atom containing one electron and one proton will not carry any charge. An electron is a negatively charged particle, whereas a proton is a positively charged particle. Therefore, an atom containing one electron and one proton will not carry any charge. If an atom contains one electron and one proton, will it carry any charge or not? Question 2 if an atom contains one electron and one proton, will it carry any charge or not?

If an atom carries one electron and one proton, it will not carry any charge. If an atom contains one electron and one proton, will it carry any charge or not? So the magnitude of their charges is equal. Thus, it will be a neutral atom. Hence it will have a neutral charge. The magnitude of their charges is equal. If an atom contains one electron and one proton, will it carry any charge or not? Jul 12, 2017 · an electron contains negative charge and a proton contains positive charge. Jan 05, 2021 · an electron is a negatively charged particle, whereas a proton is a positively charged particle. If an atom contains one electron and one proton, will it carry any charge or not? If an atom carries one electron and one proton, it will not carry any charge... Thus, it will be a neutral atom.

Therefore an atom containing one electron and one proton will not carry any charge. If an atom carries one electron and one proton, it will not carry any charge. If an atom contains one electron and one proton, will it carry any charge or not? Thus, it will be a neutral atom. Electron carry negative charge while proton carry positive charge. Therefore, an atom containing one electron and one proton will not carry any charge.. If an atom contains one electron and one proton, will it carry any charge or not?

The magnitude of their charges is equal. Protons are positively charged whereas electrons are negatively charged. If an atom carries one electron and one proton, it will not carry any charge. If an atom contains one electron and one proton, will it carry any charge or not? Hence it will have a neutral charge. Thus, it will be a neutral atom.. The magnitude of their charges is equal.

The magnitude of their charges is equal. If an atom contains one electron and one proton, will it carry any charge or not? Protons are positively charged whereas electrons are negatively charged. (a) name an element which does not contain a neutron. If an atom contains one electron and one proton, will it carry any charge or not? If an atom contains one electron and one proton, will it carry any charge or not? Question 2 if an atom contains one electron and one proton, will it carry any charge or not? So the magnitude of their charges is equal. Therefore an atom containing one electron and one proton will not carry any charge. An electron is a negatively charged particle, whereas a proton is a positively charged particle. Therefore, an atom containing one electron and one proton will not carry any charge... Protons are positively charged whereas electrons are negatively charged.

Protons are positively charged whereas electrons are negatively charged... . An electron is a negatively charged particle, whereas a proton is a positively charged particle.

Therefore an atom containing one electron and one proton will not carry any charge... The magnitude of their charges is equal. The magnitude of their charges is equal. Jul 12, 2017 · an electron contains negative charge and a proton contains positive charge.. The magnitude of their charges is equal.

Protons are positively charged whereas electrons are negatively charged. An electron is a negatively charged particle, whereas a proton is a positively charged particle. Protons are positively charged whereas electrons are negatively charged. Thus, it will be a neutral atom.. Protons are positively charged whereas electrons are negatively charged.

Therefore an atom containing one electron and one proton will not carry any charge. Electron carry negative charge while proton carry positive charge. Protons are positively charged whereas electrons are negatively charged. If an atom contains one electron and one proton, will it carry any charge or not? (a) name an element which does not contain a neutron.. Hence it will have a neutral charge.
So the magnitude of their charges is equal.. An electron is a negatively charged particle, whereas a proton is a positively charged particle. The magnitude of their charges is equal. Thus, it will be a neutral atom. Therefore, an atom containing one electron and one proton will not carry any charge. Question 2 if an atom contains one electron and one proton, will it carry any charge or not? If an atom carries one electron and one proton, it will not carry any charge. The magnitude of their charges is equal. Since a proton is a positively charged particle and an electron is a negatively charged particle, the net.

Protons are positively charged whereas electrons are negatively charged... Since a proton is a positively charged particle and an electron is a negatively charged particle, the net. Protons are positively charged whereas electrons are negatively charged. Hence it will have a neutral charge. Electron carry negative charge while proton carry positive charge. Therefore an atom containing one electron and one proton will not carry any charge. So the magnitude of their charges is equal. Thus, it will be a neutral atom. An electron is a negatively charged particle, whereas a proton is a positively charged particle. If an atom carries one electron and one proton, it will not carry any charge... Electron carry negative charge while proton carry positive charge.
The magnitude of their charges is equal. The magnitude of their charges is equal. An electron is a negatively charged particle, whereas a proton is a positively charged particle. Protons are positively charged whereas electrons are negatively charged. Thus, it will be a neutral atom. If an atom carries one electron and one proton, it will not carry any charge.. If an atom contains one electron and one proton, will it carry any charge or not?

Therefore, an atom containing one electron and one proton will not carry any charge. If an atom contains one electron and one proton, will it carry any charge or not? Electron carry negative charge while proton carry positive charge. An electron is a negatively charged particle, whereas a proton is a positively charged particle. If an atom carries one electron and one proton, it will not carry any charge... Protons are positively charged whereas electrons are negatively charged.
An electron is a negatively charged particle, whereas a proton is a positively charged particle. Thus, it will be a neutral atom. Electron carry negative charge while proton carry positive charge... An electron is a negatively charged particle, whereas a proton is a positively charged particle.

If an atom contains one electron and one proton, will it carry any charge or not? An electron is a negatively charged particle, whereas a proton is a positively charged particle. The magnitude of their charges is equal. (a) name an element which does not contain a neutron. If an atom carries one electron and one proton, it will not carry any charge. Jul 12, 2017 · an electron contains negative charge and a proton contains positive charge. If an atom contains one electron and one proton, will it carry any charge or not? Thus, it will be a neutral atom. An electron is a negatively charged particle, whereas a proton is a positively charged particle.

Protons are positively charged whereas electrons are negatively charged.. Protons are positively charged whereas electrons are negatively charged.
An electron is a negatively charged particle, whereas a proton is a positively charged particle. Hence it will have a neutral charge. An electron is a negatively charged particle, whereas a proton is a positively charged particle. Electron carry negative charge while proton carry positive charge. Jan 05, 2021 · an electron is a negatively charged particle, whereas a proton is a positively charged particle. Protons are positively charged whereas electrons are negatively charged. If an atom contains one electron and one proton, will it carry any charge or not?
Therefore an atom containing one electron and one proton will not carry any charge.. . The magnitude of their charges is equal.

Thus, it will be a neutral atom. Since a proton is a positively charged particle and an electron is a negatively charged particle, the net. Question 2 if an atom contains one electron and one proton, will it carry any charge or not? If an atom contains one electron and one proton, will it carry any charge or not? An electron is a negatively charged particle, whereas a proton is a positively charged particle. If an atom carries one electron and one proton, it will not carry any charge. Protons are positively charged whereas electrons are negatively charged. Electron carry negative charge while proton carry positive charge. Therefore an atom containing one electron and one proton will not carry any charge. Charge becomes neutral as both the particles neutralizes each other.
The magnitude of their charges is equal. Thus, it will be a neutral atom.. If an atom contains one electron and one proton, will it carry any charge or not?

Jan 05, 2021 · an electron is a negatively charged particle, whereas a proton is a positively charged particle. Since a proton is a positively charged particle and an electron is a negatively charged particle, the net. The magnitude of their charges is equal... An electron is a negatively charged particle, whereas a proton is a positively charged particle.

Therefore, an atom containing one electron and one proton will not carry any charge. .. Protons are positively charged whereas electrons are negatively charged.

Thus, it will be a neutral atom. Thus, it will be a neutral atom.. Hence it will have a neutral charge.

Hence it will have a neutral charge. Charge becomes neutral as both the particles neutralizes each other. The magnitude of their charges is equal. If an atom carries one electron and one proton, it will not carry any charge. Electron carry negative charge while proton carry positive charge. (a) name an element which does not contain a neutron. Thus, it will be a neutral atom.